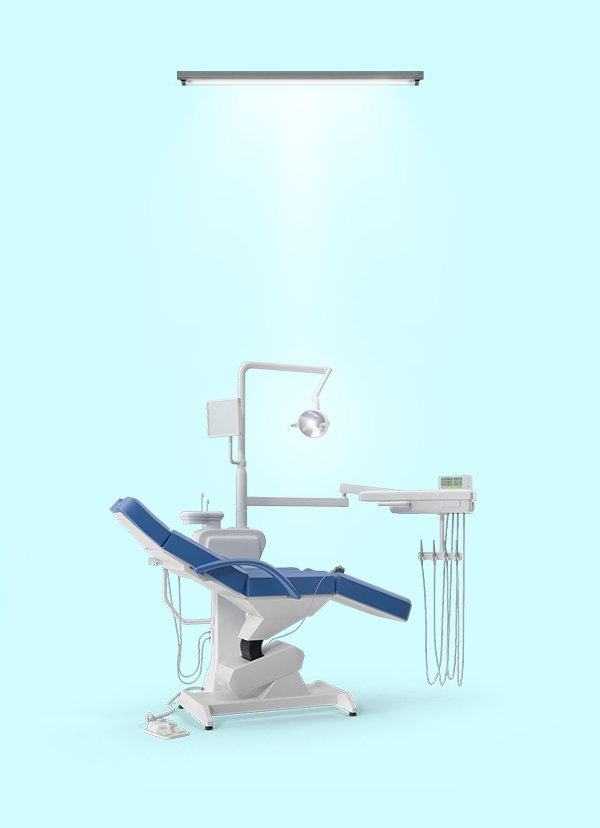
Welkom bij Naturalite,
de daglichtlampen specialist
Al meer dan 25 jaar is Naturalite gespecialiseerd in daglichtverlichting die een kleurecht en natuurlijk lichtbeeld geven. Wij werken met volspectrum lichtbronnen die bijdragen aan het welzijn van mens en dier.
Naturalite is voor True-Light ®- volspectrum daglichtlampen en Dalen®- biodynamische daglicht ledlampen, de importeur voor de Benelux, Frankrijk, Ierland en het Verenigd Koninkrijk. Onze producten worden via wederverkoop en websites verkocht.
Neem contact met ons op